Proven efficacy in both laboratory studies and real-world settings
ActivePure Technology has been proven against a wide range of contaminants
In laboratory and real-world applications, ActivePure devices have demonstrated effectiveness in helping reduce contaminants. The degree of effectiveness is impacted by conditions of the space including airflows, HVAC systems, ambient air temperatures and humidities, occupant density, VOC gas concentrations, contaminant types and concentrations, and the ActivePure device used.
Testing done on multiple devices with ActivePure Technology, excluding the ActivePure Medical Guardian, at multiple 3rd party labs. Please contact us for specific testing results and/or additional testing information.
ActivePure Medical Guardian testing for FDA Class II Clearance
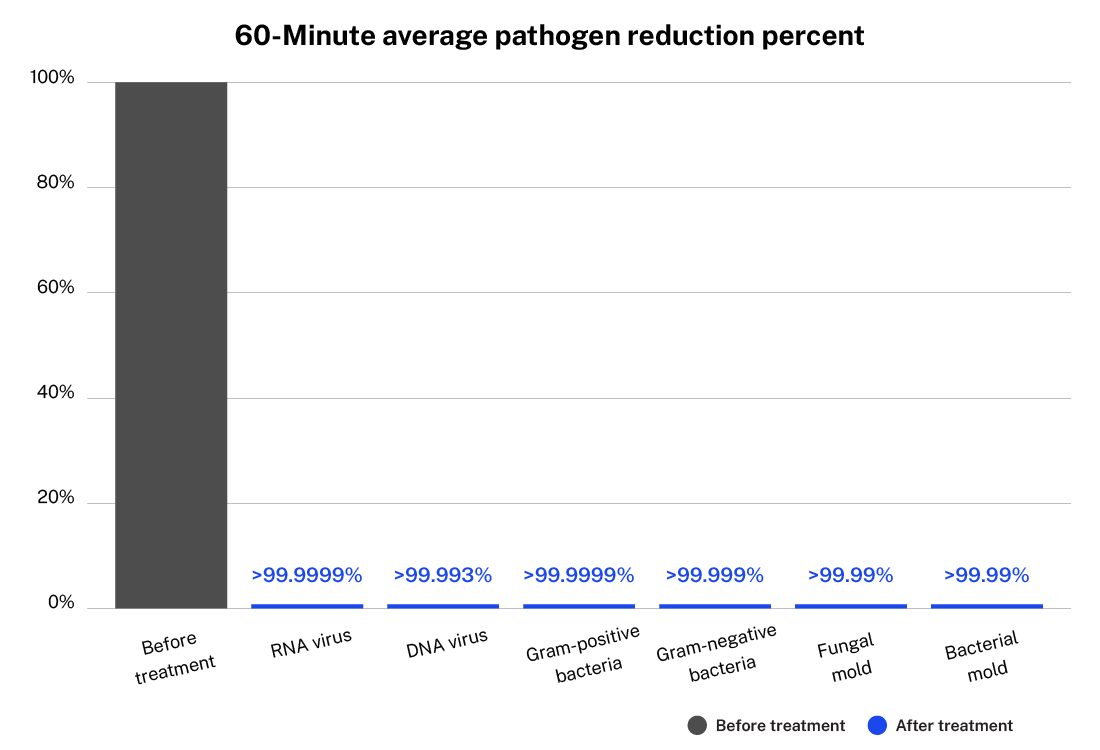
60-Minute average pathogen reduction percent
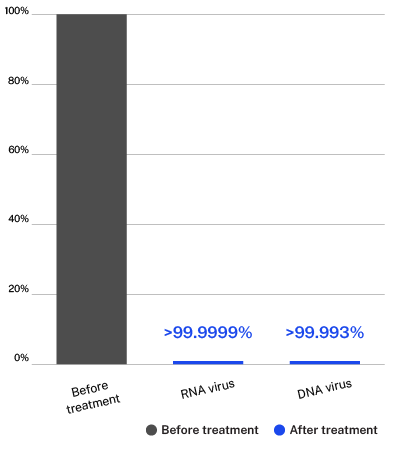
Testing done on the ActivePure Medical Guardian at Aerosol Research and Engineering Laboratories, a 3rd-party unaffiliated laboratory with no connection to ActivePure or its affiliates. RNA Virus: MS2 Bacteriophage. DNA Virus: Phi-X174 Bacteriophage.
60-Minute average pathogen reduction percent
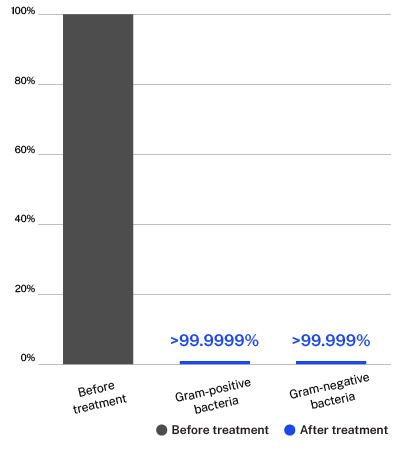
Testing done on the ActivePure Medical Guardian at Aerosol Research and Engineering Laboratories, a 3rd-party unaffiliated laboratory with no connection to ActivePure or its affiliates. Gram-positive Bacteria: Staphylococcus Epidermidis. Gram-negative Bacteria: Erwinia Herbicola.
60-Minute average pathogen reduction percent
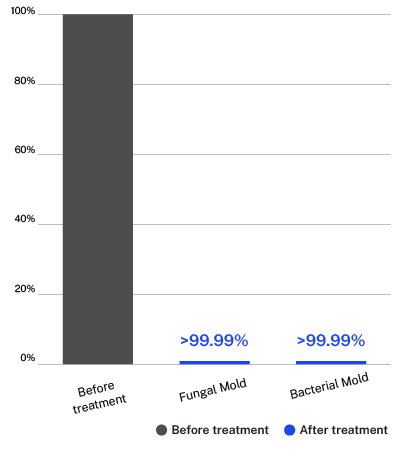
Testing done on the ActivePure Medical Guardian at Aerosol Research and Engineering Laboratories, a 3rd-party unaffiliated laboratory with no connection to ActivePure or its affiliates. Fungal Mold: Apergillus Niger. Bacterial Mold: Bacillus Globigii.
Testing done on the ActivePure Medical Guardian at Aerosol Research and Engineering Laboratories, a 3rd-party unaffiliated laboratory with no connection to ActivePure or its affiliates. RNA Virus: MS2 Bacteriophage. DNA Virus: Phi-X174 Bacteriophage. Gram-positive Bacteria: Staphylococcus epidermidis. Gram-negative Bacteria: Erwinia herbicola. Fungal Mold: Aspergillus niger. Bacterial Mold: Bacillus globigii.
Peer-reviewed science
ActivePure is committed to the highest scientific standards, with multiple published peer-reviewed studies and more in progress. The first was a study published in the American Journal of Infection Control (AJIC) on healthcare-associated infection (HAI) reduction. A summary of those results is available here.
The second study was also published by AJIC and focused on ActivePure’s efficacy against SARS-CoV-2, respiratory syncytial virus (RSV), and monkeypox virus. The results of the study highlight ActivePure’s potential as a valuable tool for improving indoor air quality and infection control. A summary of those results is available here.
Our third published peer-reviewed study was published in PLOS ONE. It examined the impact of ActivePure Technology on lab mice using the same concentrations of ActivePure proven effective against viral, bacterial, and fungal pathogens. This study showed that there were no statistically significant differences between the test group and control group due to exposure to ActivePure. A summary of those results is available here.